Retatrutide: What the Research Shows
Dr. Sieglinde Klaus
Scientific Editorial Team — Bergdorf Bioscience

Table of Contents
- 01What is retatrutide and how does it differ from semaglutide?
- 02How does triple agonism work?
- 03What does the Phase 2 study show on weight loss?
- 04What are the results for type 2 diabetes?
- 05What does research show on fatty liver disease (MASLD)?
- 06How does body composition change?
- 07What dosages have been clinically tested?
- 08Frequently Asked Questions (FAQ)
- Is retatrutide approved for therapeutic use?
- How does retatrutide differ from tirzepatide?
- How should retatrutide be stored correctly?
- What Phase 3 studies are currently running for retatrutide?
- 09Disclaimer
- 10Sources and Attribution
Retatrutide (LY3437943) is the first triple agonist that simultaneously activates GIP, GLP-1, and glucagon receptors. This research guide summarizes the current state of clinical evidence, explains the mechanism of action, and provides practical information on dosing, storage, and quality criteria for this research peptide.
What is retatrutide and how does it differ from semaglutide?
Retatrutide (LY3437943) is a peptide developed by Eli Lilly that, as a single molecule, activates three incretin and glucagon receptors: GIP receptor (GIPR), GLP-1 receptor (GLP-1R), and glucagon receptor (GCGR). This sets it apart from semaglutide (pure GLP-1 agonist) and tirzepatide (dual GIP/GLP-1 agonist). The additional glucagon receptor activation stimulates hepatic lipid oxidation and increases resting energy expenditure – a mechanism that dual agonists lack. The molecular structure consists of 36 amino acids with specific modifications: an alpha-aminoisobutyric acid (Aib) at position 2 protects against DPP-4 degradation, while a C20 fatty acid chain enables albumin binding, resulting in a plasma half-life of approximately 6 days – compatible with once-weekly administration (Urva et al., 2022).
How does triple agonism work?
The therapeutic advantage of retatrutide is based on the synergistic interplay of three signaling pathways. GLP-1 receptor: Promotes glucose-dependent insulin secretion, inhibits glucagon release, and slows gastric emptying, leading to reduced caloric intake. GIP receptor: Enhances insulin sensitivity in adipose tissue and works additively with GLP-1 to potentiate weight loss. Glucagon receptor: Stimulates hepatic lipid oxidation and thermogenesis, directly increasing energy expenditure. The meta-analysis by Wang et al. (2025) confirms that this triple activation achieves significantly higher efficacy than dual agonism alone. An important detail: the glucagon component is decisive for the pronounced effects on liver fat, as it directly addresses hepatic lipid metabolism.

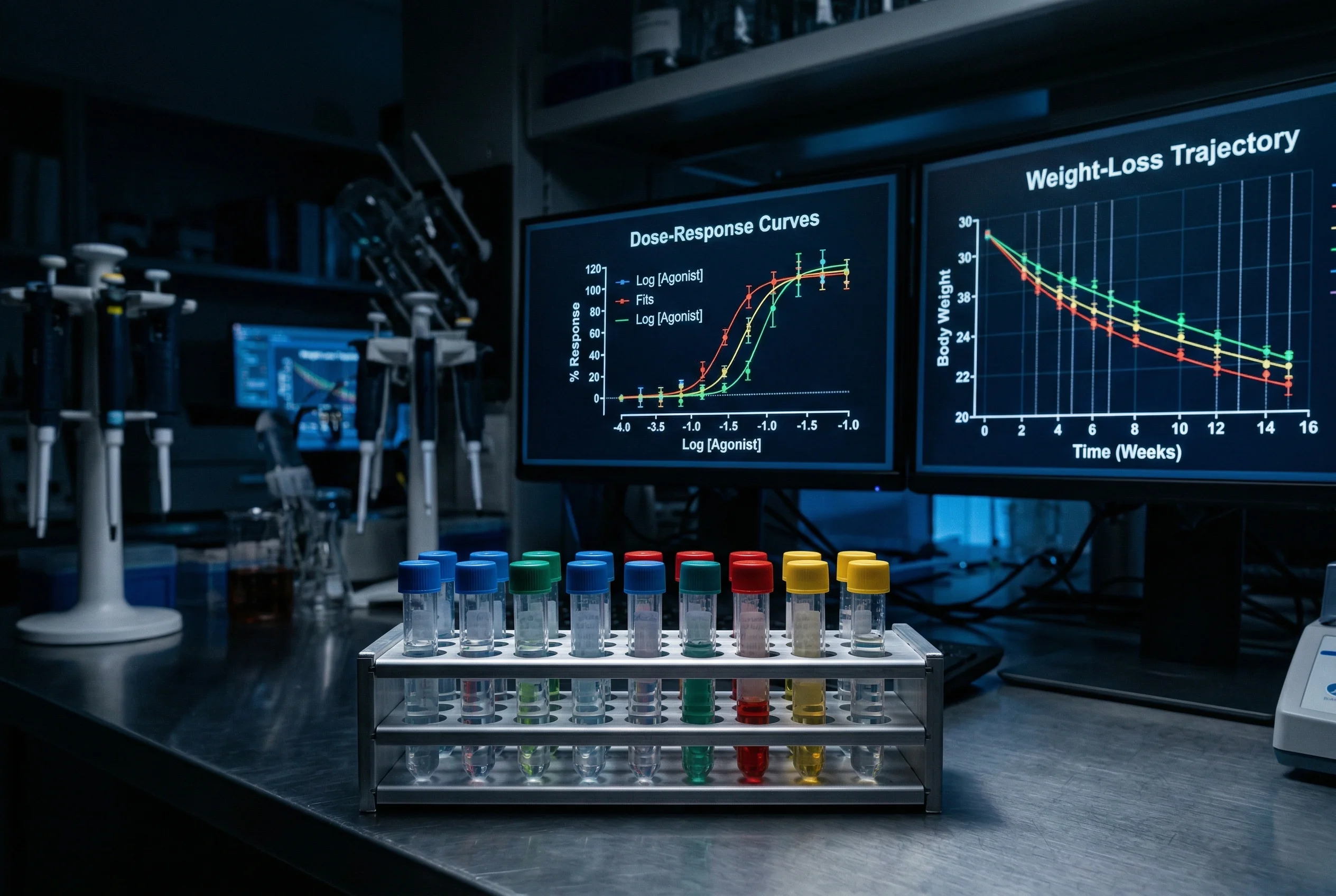
What does the Phase 2 study show on weight loss?
The pivotal efficacy study was published by Jastreboff et al. (2023) in the New England Journal of Medicine. This randomized, double-blind, placebo-controlled trial enrolled 338 adults with obesity (BMI ≥30) but without diabetes. After 48 weeks of subcutaneous injection, the following dose-dependent weight reductions were demonstrated: placebo −2.1%, 1 mg −8.7%, 4 mg −17.1%, 8 mg −22.8%, 12 mg −24.2%. These results significantly surpassed all previously published pharmacological data. For comparison: semaglutide 2.4 mg achieved approximately −14.9% in the STEP-1 study, and tirzepatide 15 mg reached up to −22.5% in SURMOUNT-1. The most common side effects were gastrointestinal in nature (nausea, diarrhea, vomiting), predominantly occurring during the dose escalation phase and generally mild to moderate in severity (Jastreboff et al., 2023).
What are the results for type 2 diabetes?
In parallel with the obesity study, Rosenstock et al. (2023) investigated the efficacy and safety of retatrutide in adults with type 2 diabetes. This 36-week Phase 2 study tested doses from 0.5 mg to 12 mg. The results: HbA1c reduction of up to −2.02 percentage points (12 mg), with 71% of participants in the highest dose group reaching the target of <5.7%. Body weight decreased dose-dependently by up to −16.9%. Additionally, significant improvements in fasting glucose, triglycerides, and systolic blood pressure were documented. These data demonstrate that retatrutide addresses not just body weight, but the full spectrum of cardiometabolic risk factors (Rosenstock et al., 2023).
What does research show on fatty liver disease (MASLD)?
Metabolic dysfunction-associated steatotic liver disease (MASLD, formerly known as NAFLD) affects an estimated 30% of the global population and is closely linked to obesity and insulin resistance. The Phase 2a study by Hartman et al. (2024), published in Nature Medicine, investigated retatrutide in 98 participants with MASLD. The results were striking: at the 12 mg dose, liver fat was reduced by 86%, and 93% of participants achieved normal liver fat content (<5%). This effect significantly exceeds the results of pure GLP-1 agonists in this indication. The glucagon component is responsible for this, as it directly stimulates hepatic fatty acid oxidation – a mechanism unique to the triple agonist approach (Hartman et al., 2024).
How does body composition change?
A frequent criticism of pharmacologically induced weight loss is the potential loss of lean mass (muscle, bone). The DXA substudy of the Phase 2 trial addressed this concern. It showed that approximately 74% of the weight lost with retatrutide came from fat mass, while lean mass loss was proportionally comparable to other weight loss interventions. In the 12 mg group, total fat mass decreased by approximately 13.6 kg over 48 weeks. Visceral fat (metabolically particularly harmful) was disproportionately reduced – a pattern consistent with the glucagon-mediated hepatic lipolysis. These data suggest that retatrutide primarily addresses adipose tissue while relatively preserving lean mass (DXA substudy, 2025).
What dosages have been clinically tested?
In the Phase 1b study by Urva et al. (2022), single doses of 0.5 mg to 12 mg and multiple doses up to 12 mg were investigated. The pharmacokinetics were dose-proportional with a half-life of approximately 6 days and steady-state achievement after 4–5 weeks. In the Phase 2 studies, the following dose ranges were tested: Obesity: 1, 4, 8, and 12 mg weekly. Type 2 Diabetes: 0.5, 4, 8, and 12 mg weekly. MASLD: 1, 4, 8, and 12 mg weekly. All regimens used a step-up titration protocol (gradual dose increase over several weeks) to minimize gastrointestinal side effects. The 12 mg dose consistently showed the highest efficacy across all endpoints. For research purposes, Bergdorf Bioscience offers retatrutide in vial sizes from 10 mg to 50 mg.
Frequently Asked Questions (FAQ)
Is retatrutide approved for therapeutic use?
No. Retatrutide (LY3437943) is currently in Phase 3 clinical development at Eli Lilly. It is exclusively available as a research peptide and is not approved for human consumption. Approval is not expected before 2027/2028.
How does retatrutide differ from tirzepatide?
Tirzepatide (Mounjaro®/Zepbound®) is a dual GIP/GLP-1 agonist, while retatrutide additionally activates the glucagon receptor. This third component drives hepatic lipid oxidation and directly increases energy expenditure. In clinical comparisons, retatrutide achieved higher weight reduction (24.2% vs. 22.5% after 48 weeks) and significantly greater liver fat reduction.
How should retatrutide be stored correctly?
As lyophilized powder, retatrutide should be stored at 2–8 °C (refrigerator), protected from light and moisture. For long-term storage (>3 months), −20 °C is recommended. After reconstitution with bacteriostatic water: 2–8 °C, use within 28 days. Repeated freeze-thaw cycles should be avoided.
What Phase 3 studies are currently running for retatrutide?
Eli Lilly is conducting several Phase 3 studies under the TRIUMPH program name. These include trials for obesity, type 2 diabetes, MASLD, and knee osteoarthritis. The first results (TRIUMPH-4) were published in December 2025, showing 28.7% mean weight loss at 68 weeks. Seven further studies are expected to complete throughout 2026.
Disclaimer
Important notice: Retatrutide is a research peptide and not intended for therapeutic use in humans. All information in this guide is based on published scientific studies and serves exclusively for informational purposes. Bergdorf Bioscience supplies retatrutide solely for research purposes. Any application beyond in vitro or preclinical research is the sole responsibility of the researcher. We expressly advise against self-medication.
Sources and Attribution
This guide was prepared on March 22, 2026 by the scientific editorial team at BergdorfBio. All data are based on peer-reviewed publications with DOI-verified links:
- Urva et al. (2022) — Lancet; DOI: 10.1016/S0140-6736(22)02033-5
- Jastreboff et al. (2023) — NEJM; DOI: 10.1056/NEJMoa2301972
- Rosenstock et al. (2023) — Lancet; DOI: 10.1016/S0140-6736(23)01053-X
- Hartman et al. (2024) — Nature Medicine; DOI: 10.1038/s41591-024-03018-2
- DXA-Substudie (2025) — Lancet Diabetes & Endocrinology; DOI: 10.1016/S2213-8587(25)00092-0
- Meta-Analyse (2025) — Expert Rev Clin Pharmacol; DOI: 10.1080/17512433.2025.2450254
Last updated: March 22, 2026. For questions or comments, please contact our scientific team via the contact form.
